 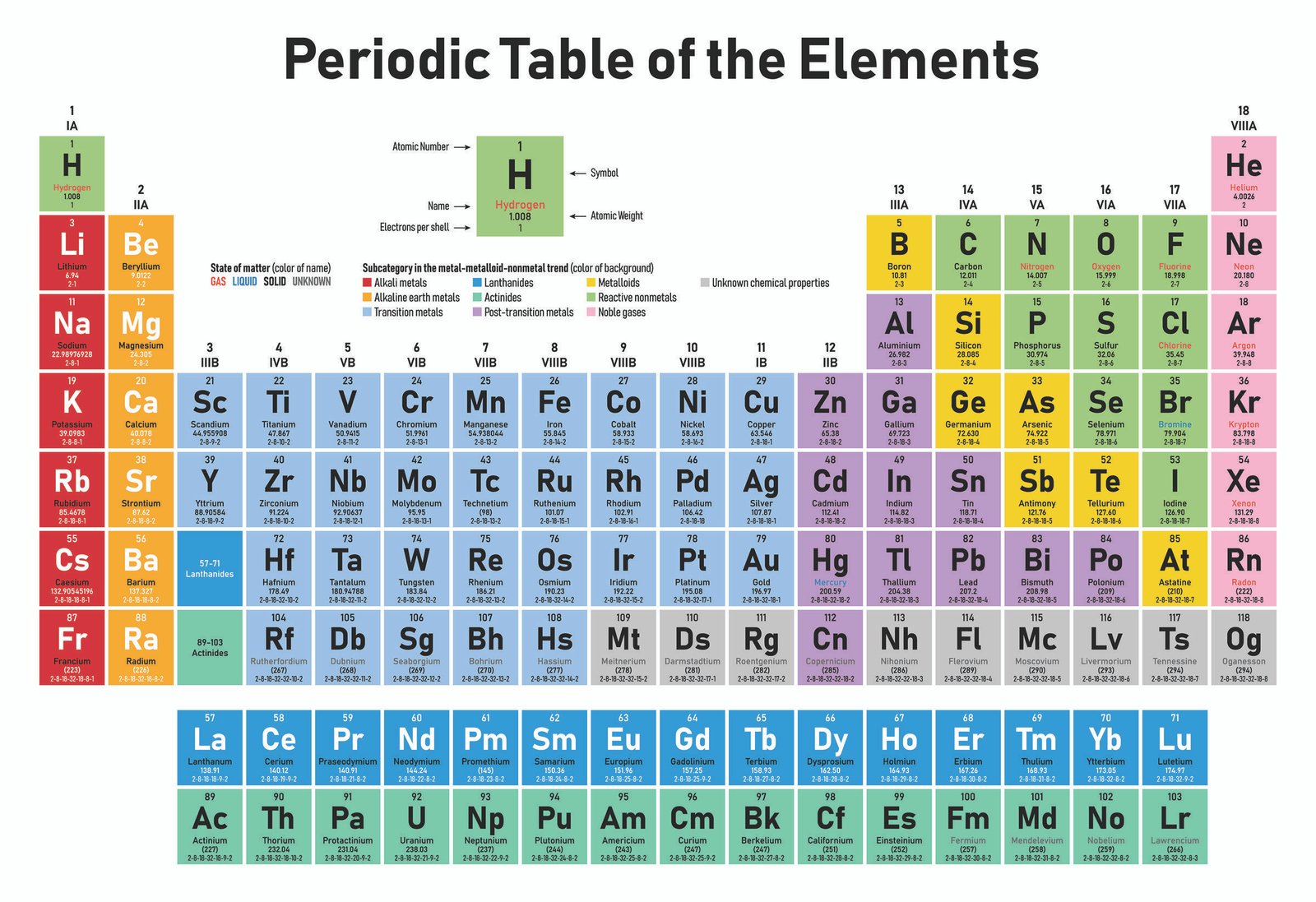 
After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. 
This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. 180 Cards (9 cards for each of the first 20 elements) in color and blackline.The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital.Store them using a method that works for you so you can use them from year-to-year.The cost of a corner punch is a great investment. It takes a little more time, but the added professional look of rounding the corners is worth it.This reduces the number of cuts and eliminates waste. The cards are designed to be printed to 100% and fill an entire 8 ½” x 11” sheet of paper. It also dramatically increases the useful life of the cards. While this is not strictly necessary, the cards “deal” easier when laminated. You can use one color per deck to distinguish between decks, or you can print each deck on a variety of colors. If you are printing the blackline deck, I recommend printing on colored card stock. If you are printing the color deck, I recommend using white card stock. However, when using the full deck of 180 cards, one deck is more than enough to keep 9 students practicing their periodic table skills. In the beginning, when introducing new topics and using fewer cards, you may need more decks. If color printing is not an option blackline option look great on colored cardstock. The color cards are fun and look very professional. Decide whether you are going to print in color or blackline.There are so many ways to use these cards, you will used them over and over again. Assign the atomic number and atomic mass cards or the protons, electrons and neutrons cards and have the students identify the element. Give your students the name and symbol cards and have them match them together. 
Give your students just the symbol cards and have them write down the names. When first introducing the periodic table and the necessary skills to use it, these cards can be used as simple task cards or a card sort.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
